All About Access
A leading global provider of vascular and other access products for a range of interventional and endovascular procedures.
About Galt OEM...
Galt Medical Corp. has been a leading provider of vascular and interventional medical devices for over 30 years, developing and manufacturing a range of products for interventional procedures. Galt offers products private labeled to medical device OEMs and Galt branded to clinicians worldwide through a network of distributors. Their regulatory, customer service, engineering, and marketing teams are located in our Garland, Texas headquarters, with manufacturing done both at our Garland location and in Costa Rica. Clinicians in six continents use Galt products for a range of Interventional Radiology, Interventional Cardiology, Vascular Surgery, and IV Therapy procedures.
Galt provides custom products and associated design services to a range of large and small medical device companies. Projects range from exclusive, minor modifications of existing Galt products for an OEM partner, to unique products based on an OEM’s requirements or specifications. Our manufacturing capabilities include, but are not limited to: tipping, forming, shaping, injection molding, hot stamping, hole punching, markings, assembly, packaging, tooling, and rapid prototyping/3D printing. Galt also provides regulatory support to many of its contract manufacturing customers.

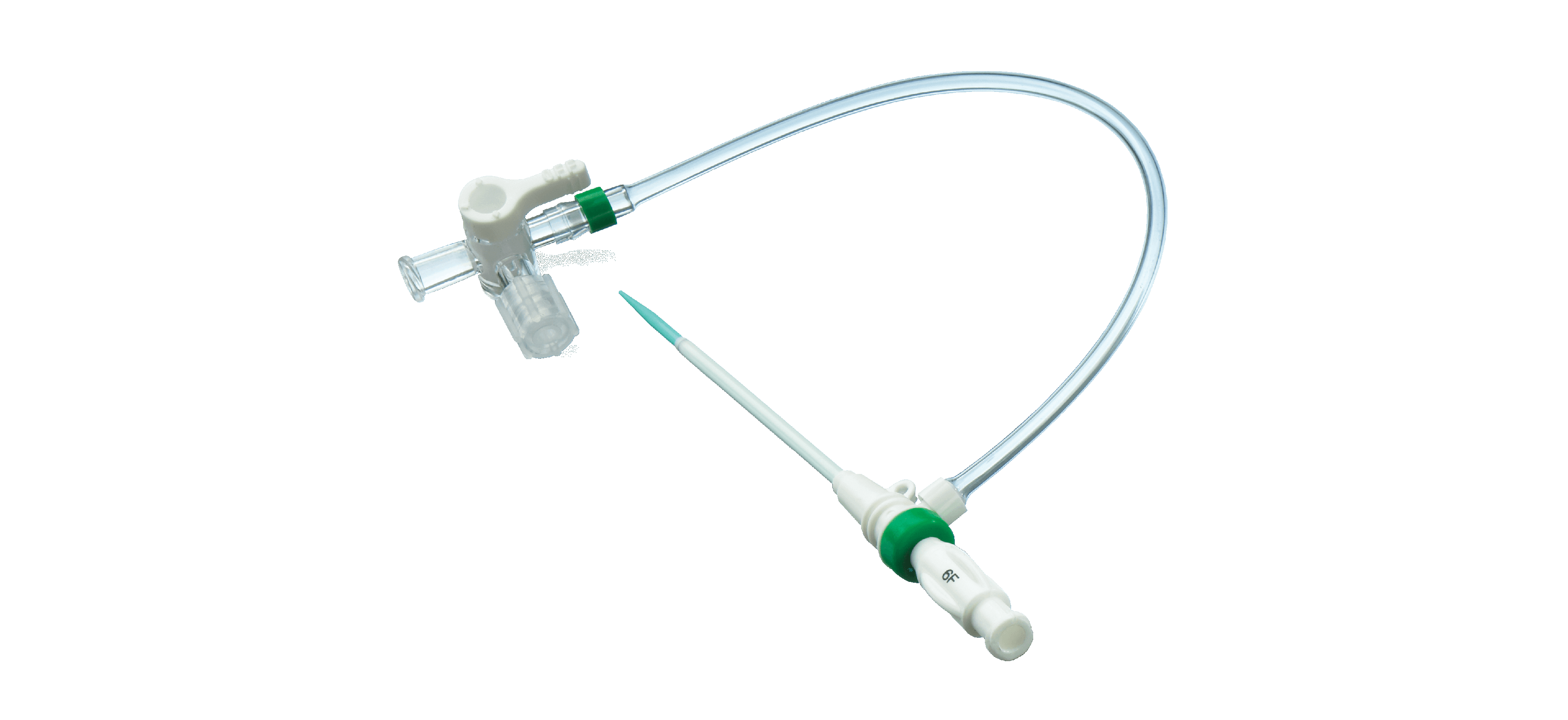
Products
From specialty guidewires to micro, tearaway and valve introducers, Galt Medical Corp. provides innovative devices to aid in an efficient and successful procedure. From full kits to individual components, we have the right offering for you.
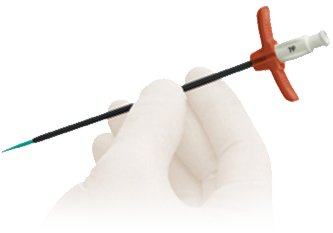
OEM Solutions
We offer flexible product configuration options to meet your needs. Leveraging our medical device development engineering and extensive manufacturing resources allows our OEM partners to expedite time to market.

Trusted Experience
Galt Medical Corp. has been a leader in designing, developing, manufacturing and marketing disposable medical devices for over 3 decades.
GALT OEM's Product Categories
Become a connection on LinkedIn.